Demonstrated safety profile for REXULTI® (brexpiprazole) in adults
Study 3 and Study 4: REXULTI—adverse reactions that occurred in ≥2%
of patients and greater incidence vs placebo from two 6-week pivotal trials
Study 3 and Study 4: REXULTI—
adverse reactions that occurred in ≥2%
of patients and greater incidence vs placebo from
two 6-week pivotal trials
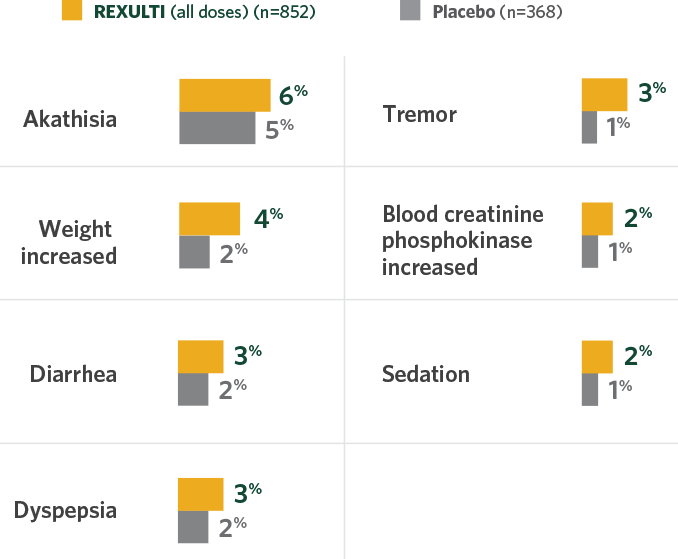
- Most common adverse reaction occurring in ≥4% of patients and at least twice the rate of placebo was weight increased
- In both trials, akathisia occurred most often during the first 3 weeks of treatment, and no incidences resulted in treatment discontinuation1,2
- The safety population included patients receiving 1 mg/day
Additional safety information across 2 pivotal trials
REXULTI—safety considerations evaluated over 6 weeks3

QTc, corrected QT.
REXULTI: Metabolic data across all doses in short- and long-term trials in adults
WEIGHT CHANGE
Weight change results across all doses4,5
aAfter the conclusion of the 6-week trial, patients who elected to remain in the trial, patients from the Phase 2 trial, and de novo patients were treated with flexible doses of REXULTI (1 mg to 6 mg) in a 52-week, open-label trial, and no placebo control was included. Safety data were collected at regular intervals throughout the 52 weeks. The overall N was 813; n values for individual parameters varied.6
bSD for mean weight increased at 6 weeks were 3.4 kg for REXULTI and 2.7 kg for placebo, at 26 weeks was 5.5 kg for REXULTI, and at 52 weeks was 7.3 kg for REXULTI.6,7
The standard conversion for kilograms (kg) to pounds (lb) is 1 kg for 2.20462 lb.
SD, standard deviation.
Discontinuation due to weight increased across all doses6,8
aAfter the conclusion of the 6-week trial, patients who elected to remain in the trial, patients from the Phase 2 trial, and de novo patients were treated with flexible doses of REXULTI (1 mg to 6 mg) in a 52-week, open-label trial, and no placebo control was included. Safety data were collected at regular intervals throughout the 52 weeks. The overall N was 813; n values for individual parameters varied.6
METABOLIC CHANGE
Percentage of patients whose values shifted from baseline to post-baseline6,9
Percentage of patients whose values shifted from baseline to post-baseline6,9
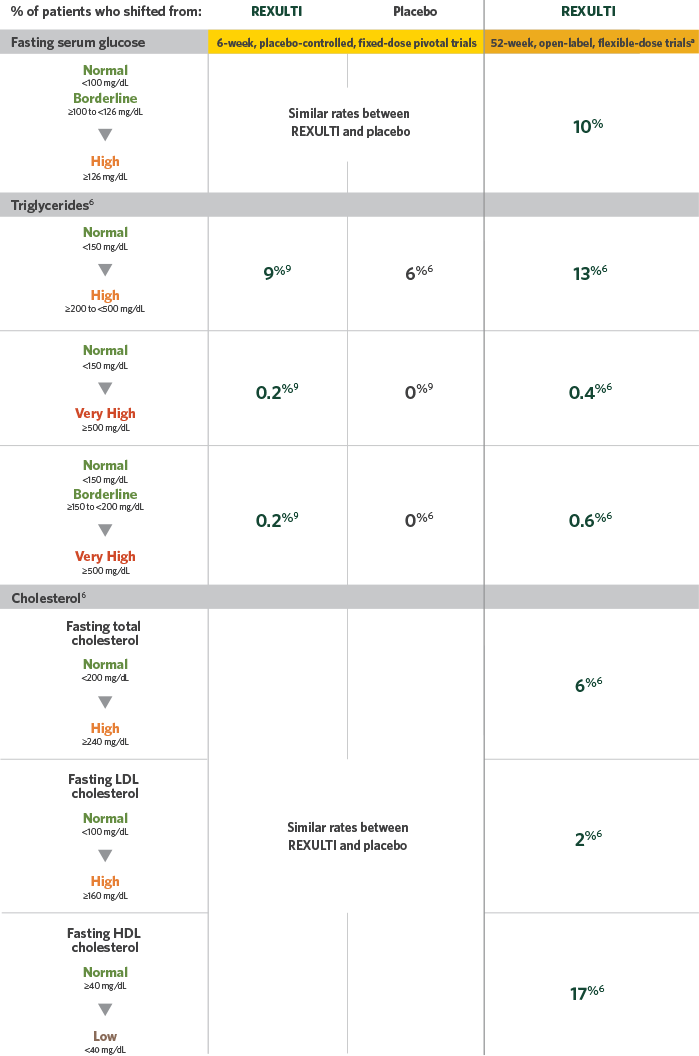
aAfter the conclusion of the 6-week trial, patients who elected to remain in the trial, patients from the Phase 2 trial, and de novo patients were treated with flexible doses of REXULTI (1 mg to 6 mg) in a 52-week, open-label trial, and no placebo control was included. Safety data were collected at regular intervals throughout the 52 weeks. The overall N was 813; n values for individual parameters varied.6
HDL, high-density lipoprotein; LDL, low-density lipoprotein.
Important Warning and Precaution for Metabolic Changes
Atypical antipsychotic drugs have been associated with metabolic changes that include Hyperglycemia/Diabetes Mellitus, Dyslipidemia, and Weight Gain. Clinical monitoring is recommended.
